Ordering
*link will take you to our exclusive distribution partner site
*link will take you to our exclusive distribution partner site
BIOTAQ is a highly purified, thermostable DNA polymerase offering high yield over a wide range of PCR templates and a good choice for routine PCR assays.
BIOTAQ is a robust preparation and delivers high yields with minimal background. BIOTAQ possesses 5´-3´ exonuclease activity and leaves an ´A´ overhang such that the PCR product is suitable for effective integration into TA cloning vectors.
BIOTAQ™ is a purified thermostable DNA polymerase offering high yield over a wide range of PCR templates, and is a good choice for routine assays. BIOTAQ is a robust preparation and delivers high yields with minimal background. BIOTAQ possesses 5´-3´ exonuclease activity and leaves an ´A´ overhang such that the PCR product is suitable for effective integration into TA cloning vectors.
BIOTAQ is supplied with 10x NH4-based Reaction Buffer, which provides optimal conditions for most experiments. Additional MgCl2 is provided to allow reaction conditions to be adjusted to suit the template.
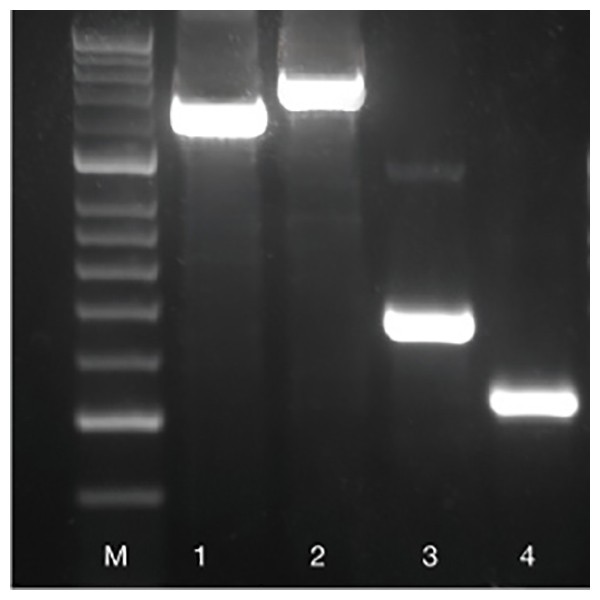
Fig. 1 Amplification of a variety of fragments.
Four different genes were amplified from mouse genomic DNA using BIOTAQ DNA Polymerase: 1.4 kb and 1.6 kb fragment of rn18s gene (lanes 1 and 2), 500 bp fragment of Fabpi gene (lane 3), 350 bp fragment of IL-2 gene (lane 4). HyperLadder 50bp (M).
|
Reagent |
500 Units |
2500 Units |
|
BIOTAQ DNA Polymerase |
1 x 100 µL |
5 x 100 µL |
|
10x NH4 Reaction Buffer |
2 x 1.2 mL |
10 x 1.2 mL |
|
50 mM MgCl2 Solution |
1 x 1.2 mL |
5 x 1.2 mL |
5 u/μL
All components should be stored at -20°C upon receipt for optimum stability. Repeated freeze/thaw cycles should be avoided.
When stored under the recommended conditions and handled correctly, full activity of the reagents is retained until the expiry date indicated on the outer box label.
On Dry Ice or Blue Ice.
One unit will incorporate 10 nmoles of dNTPs in 30 min at 72°C.
BIOTAQ is a trademark of Meridian.
| BIOTAQ, IMMOLASE, MangoTaq, MyTaq DNA Polymerase |
1.1 x 10-4 base substitutions/bp (Tindall and Kunkel, 1988) |
| 2.4 x 10-5 frameshift mutations/bp (Tindall and Kunkel, 1988) | |
| 2.1 x 10-4 errors/bp (Keohavang and Thilly, 1989) | |
| 7.2 x 10-5 errors/bp (Ling et al., 1991) | |
| 8.9 x 10-5 errors/bp (Cariello et al., 1991) | |
| 2.0 x 10-5 errors/bp (Lundberg et al., 1991) | |
| 1.1 x 10-4 errors/bp (Barnes, 1992) | |
| ACCUZYME | 1.6 x 10-6 errors/base (Lundberg et al., 1991). |
| VELOCITY | 4.4 x 10-7 errors/base (Frey & Suppmann, 1995). |
| Observation | Recommended Solution(s) |
| No or low PCR yield | Enzyme concentration too low – increase the amount of enzyme in 0.5 U increments. |
| Primers degraded – check quality and age of the primers. | |
| Magnesium concentration too low – increase concentration in 0.25 mM increments with a starting concentration of 1.75 mM. | |
| Primer concentration not optimized. Titrate primer concentration (0.3-1 µM); ensuring that both primers have the same concentration. | |
| Template concentration too low – Increase concentration of template. | |
| Perform a positive control to ensure that the enzyme, dNTPs and buffers are not degraded and/or contaminated. | |
| Multiple Bands | Primer annealing temperature too low. Increase annealing temperature. Primer annealing should be at least 5°C below the calculated Tm of primers. |
| Prepare master mixes on ice or use a heat-activated polymerase. | |
| For problems with low specificity. Try adding 3% DMSO (not supplied) to improve specificity. | |
| Smearing or artifacts | Template concentration too high. Prepare serial dilutions of template. |
| Too many cycles. Reduce the cycle number by 3-5 to remove non-specific bands. | |
| Enzyme concentration too high - decrease the amount of enzyme in 0.5 U increments. | |
| Extension time too long. Reduce extension time in 0.5-1 minute increments. |